Though not a completely rigorous explanation, you can imagine a state of a system being a way to uniquely describe the system in terms of its arrangement of particles, with an associated energy eigenvalue. However, insofar as there are multiple ways that particles can be arranged in the system that have indistinguishable observables, we call these different ways of organization "microstates". So, the particles that comprise a unique state of the system can be arranged in several basically equivalent ways.
For example, in the ground state of molecular oxygen, there are two half filled orbitals. There are two unpaired electrons, each with an associated spin of ± 1/2. The total spin magnitude is therefore 1, which allows values of 1, 0, or -1, corresponding to


,
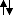
and


, respectively.* In the absence of a magnetic field, these three possible arrangements are degenerate and indisguishable from each other. You may think of it as being impossible for determine which of these three possible states the system is in at any time, if that is helpful. So we label these as microstates and call the overall state a "triplet" - there are three possible spin microstates. Bear in mind that if we introduce a magnetic field, these three configurations are no longer degenerate. In certain experiments we would see a single spectroscopic absorption line split into three separate lines in the presence of a magnetic field. This is called the Zeeman effect, and it is the origin of the term "triplet state".
*It is actually a little more complicated than this, because there is also an associated singlet state. So you actually have four total microstates, comprised of a singlet and a triplet, or four combinations:


,


,


,


. It is customary to describe the two states with opposing spins as linear combinations of each other, as depicted here:
https://en.wikipedia.org/wiki/Triplet_stateThe singlet state involves on microstate, and the triplet involves three of them.
3) for example if an atom X has this term symbol associated: 3P1
According to this,in which state is the atom??? and in which microstate/s 
The system is in a triplet state, meaning it is in a superposition of three possible degenerate eigenstates. It is not possible to experimentally distinguish which of these microstates the system is in at a given time, although the number of degenerate microstates does have statistical mechanical manifestations that can be observed experimentally. This is distinguished, say from a state labeled
1P
1, which has a completely different energy eigenvalue, and is therefore experimentally distinguishable.
Microstates associated with orbital angular momenta (the "P") don't work any differently. The three p orbitals in an isolated atom are degenerate in an isotropic field. If such an atom had a single electron, you could not tell which of the three p orbitals the electron was located in at a given time. In some interpretations of quantum mechanics, it is in all three simultaneously. Only if the degeneracy is broken does this situation change. This is the strongest distinction of "microstate" versus "state". One you can experimentally observe, the other only has a statistical mechanical relevance. Term symbols are a convenient way to keep track of all the possible microstates, and how they are grouped together in experimentally observable states of the system.